Kontakty
Rychlý kontakt (24h)
+420 543 131 111
Nový onkologický pacient
+420 543 135 809
Zdravotnická pracoviště
Pro zdravotníky a odborníky
Stáže a výuka
Mezioborové indikační komise
Úsek podpory zdravotnických týmů a komisí
Odbor nelékařských zdravotnických pracovníků
Odbor léčebně-preventivní péče
Nezdravotnická pracoviště
Vedení nemocnice
Ekonomický odbor
Oddělení personální a mzdové
Právní oddělení
Odbor informačních technologií
Odbor kanceláře ředitele
Odbor hospodářsko-technických služeb
Úsek zdravotních pojišťoven
Úsek tiskového mluvčího
Etická komise
Odbor vědy, výzkumu a vzdělávání
Centrální kartotéka a příjem
Úsek radiační ochrany
Úsek interního auditu
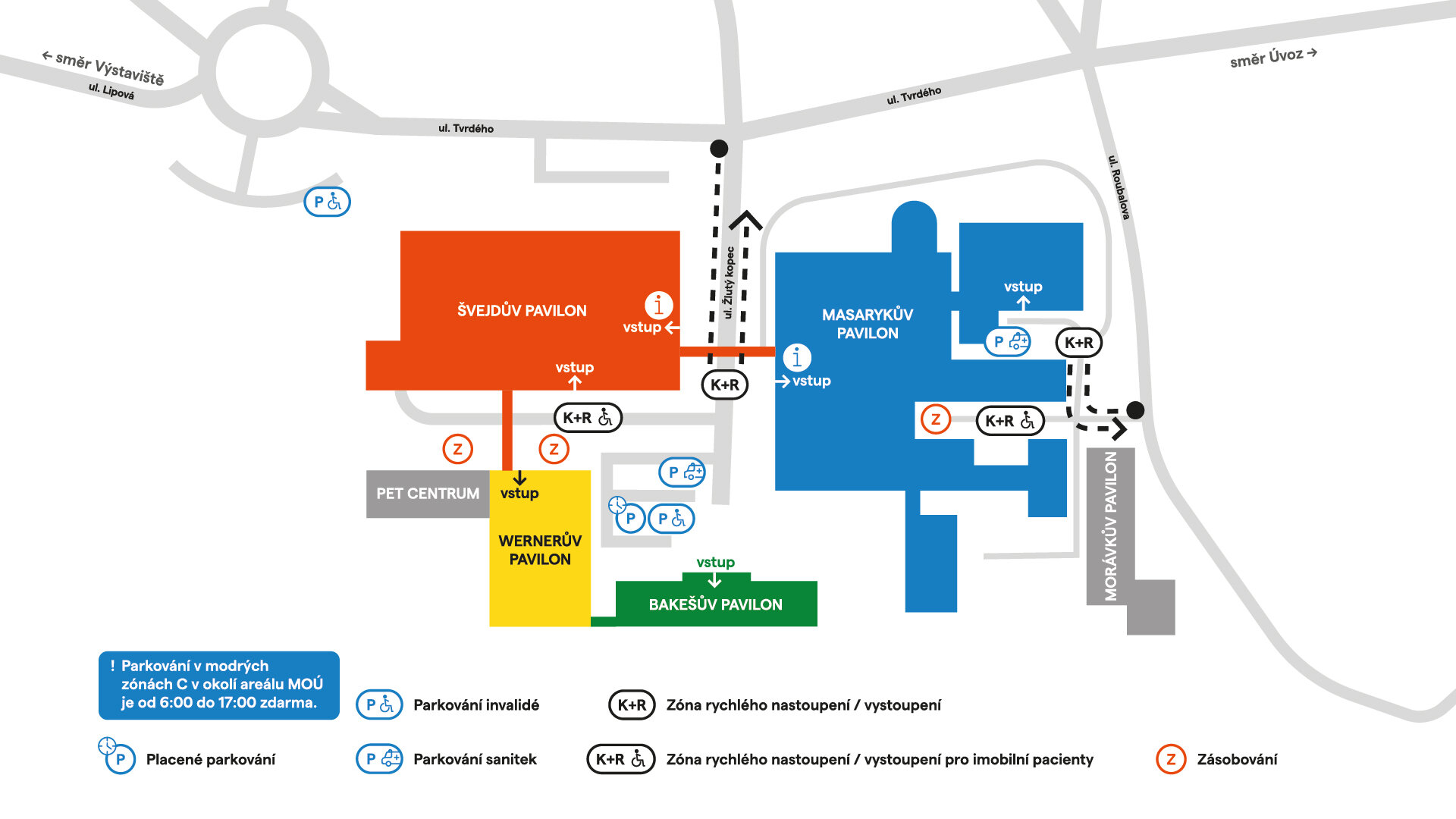
Masarykův onkologický ústav
Žlutý kopec 7
656 53 Brno
Česká republika
T +420 543 131 111
E [obfuscate_1_|98|105|109|99|90|114|64|104|109|108|44|99|117] (Kancelář ředitele)
E [obfuscate_1_|100|97|102|114|108|112|97|94|99|55|107|111|112|44|90|120] (Ekonomický odbor)
E [obfuscate_1_|109|112|96|107|55|107|111|112|44|90|120] (Oddělení personální a mzdové)
E [obfuscate_1_|99|100|112|97|92|108|116|109|115|100|62|109|106|115|37|97|122] (pro pacienty)
IČO: 00209805
DIČ: CZ00209805
Bankovní spojení: 87535621/0710
SWIFT: CNBACZPP
IBAN: CZ5807100000000087535621
Identifikátor datové schránky: 7vqnyc6
FAX: 543132455 (sekretariát KKOP)